Join CNN’s Marvel Concept science publication. Explore the universe with news on fascinating discoveries, scientific advancements and more.
CNN
—
The UK has turn out to be the primary nation to offer regulatory approval to a medical remedy involving the revolutionary CRISPR gene modifying instrument.
The nation’s Medicines and Healthcare merchandise Regulatory Company stated Thursday it had given a greenlight to a remedy often known as Casgevy, which will likely be used to deal with sickle cell illness and beta thalassemia. Each genetic situations are brought on by errors within the genes for hemoglobin, which is utilized by crimson blood cells to hold oxygen across the physique. There isn’t a identified universally profitable remedy for both dysfunction.
Sickle cell illness, which may end up in assaults of debilitating ache, is extra widespread in individuals with an African or Caribbean household background. Beta thalassemia primarily impacts individuals of Mediterranean, South Asian, Southeast Asian and Center Japanese origin, the statement said.
“Each sickle cell illness and β-thalassemia are painful, life-long situations that in some instances could be deadly. To this point, a bone marrow transplant — which should come from a intently matched donor and carries a danger of rejection — has been the one everlasting remedy choice,” stated Julian Seaside, the interim government director of healthcare high quality and entry on the MHRA, in a press release.
“I’m happy to announce that we have now authorised an modern and first-of-its-kind gene-editing remedy known as Casgevy, which in trials has been discovered to revive wholesome haemoglobin manufacturing within the majority of contributors with sickle-cell illness and transfusion-dependent β-thalassaemia, relieving the signs of illness.”
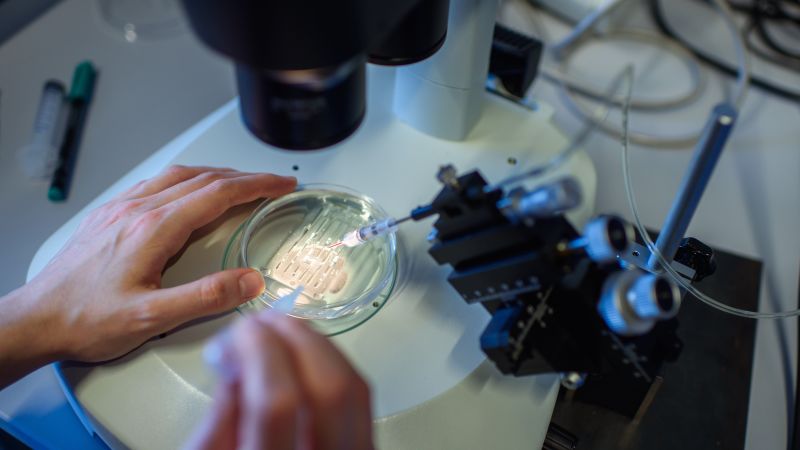
The CRISPR-Cas9 gene modifying approach permits scientists to make very exact modifications to DNA. Its inventors — Emmanuelle Charpentier and Jennifer A. Doudna — won a Nobel Prize in chemistry in 2020.
Casgevy isn’t a easy tablet or injection. The remedy, made by Vertex Pharmaceuticals, is run by taking stem cells out of a affected person’s bone marrow and modifying a gene within the cells in a lab. Sufferers then should bear a “conditioning remedy,” which may contain an immunosuppressing drug, radiotherapy or chemotherapy, to organize the bone marrow earlier than the modified cells are infused again into the affected person, in keeping with the MHRA.
“After that, sufferers could must spend at the least a month in a hospital facility whereas the handled cells take up residence within the bone marrow and begin to make crimson blood cells with the steady type of haemoglobin,” in keeping with the assertion.
The US Meals and Drug Administration is evaluating the identical remedy and is expected to make a decision on whether or not to greenlight it by December 8.
“This can be a nice step within the development of medical approaches to sort out genetic illnesses we by no means thought could be doable to treatment,” stated Alena Pance, a senior lecturer in genetics on the College of Hertfordshire, in a press release launched by the Science Media Centre, which gives info to journalists.
“Modifying the stem cells from the bone marrow of the affected person avoids the issues related to immune compatibility, i.e. trying to find donors that match the affected person and following immunosuppression, and constituting an actual treatment of the illness fairly than a remedy,” Pance added.
The discharge from the MHRA didn’t say how a lot the remedy would price, nevertheless it’s more likely to be costly.
CRISPR-Cas9 has had a significant impression on biomedical analysis, medical drugs and agriculture and is extensively utilized in labs around the globe.
The cutting-edge know-how was tainted with controversy after Chinese scientist He Jiankui introduced in 2018 he had created the world’s first gene-edited infants. Scientists say the highly effective approach shouldn’t be used to control human genes that will likely be handed down from one era to the following.